Cholesterol Metabolism/Cholesterol is an essential lipid for mammalian cells and its homeostasis is tightly regulated. Disturbance of cellular cholesterol homeostasis is linked to atherosclerosis and cardiovascular diseases. A central role in the sensing and regulation of cholesterol homeostasis is attributed to the endoplasmic reticulum (ER). This organelle harbors inactive transcription factors, which sense ER cholesterol levels and initiate transcriptional responses after activation and translocation into the nucleus. Thereupon, these responses enable adaption to high or low cellular cholesterol levels. Besides the abovementioned canonical functions, ER stress—induced by metabolic burden—and the resulting unfolded protein response influence cholesterol metabolism relevant to metabolic disorders. This review summarizes basic as well as recent knowledge on the role of the ER in terms of the regulation of cholesterol metabolism.
Lipid and Lipoprotein Metabolism
Lipids = cholesterol and triglyceride – are insoluble in plasma and are transported in lipoproteins.
Functions = energy utilization, steroid hormone production, bile acid production, lipid deposition.
Lipoprotein consists of esterified and unesterified cholesterol, triglycerides, phospholipids, and apolipoproteins. The proteins function as cofactors and ligands for receptors.
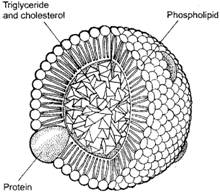
Major lipoproteins include:
- Chylomicrons – large particles that carry dietary lipid
- Very low-density lipoprotein – carry endogenous triglyceride and some cholesterol
- Intermediate density lipoprotein – carry cholesterol esters and triglycerides
- Low-density lipoprotein – carry cholesterol esters
- High-density lipoprotein – carry cholesterol esters
Exogenous pathway for lipid metabolism:
- Dietary cholesterol and fatty acids are absorbed.
- Triglycerides are formed in the intestinal cell from free fatty acids and glycerol and cholesterol is esterified.
- Triglycerides and cholesterol combine to form chylomicrons.
- Chylomicrons enter the circulation and travel to peripheral sites.
- In peripheral tissues, free fatty acids are released from the chylomicrons to be used as energy, converted to triglyceride or stored in adipose.
- Remnants are used in the formation of HDL.
Endogenous pathway for lipid metabolism:
- VLDL is formed in the liver from triglycerides and cholesterol esters.
- These can be hydrolyzed by lipoprotein lipase to form IDL or VLDL remnants.
- VLDL remnants are cleared from the circulation or incorporated into LDL.
- LDL particles contain a core of cholesterol esters and a smaller amount of triglyceride.
- LDL is internalized by hepatic and nonhepatic tissues.
- In the liver, LDL is converted into bile acids and secreted into the intestines.
- In non-hepatic tissues, LDL is used in hormone production, cell membrane synthesis, or stored.
- LDL is also taken up by macrophages and other cells which can lead to excess accumulation and the formation of foam cells which are important in plaque formation.
Cholesterol Metabolism
One-quarter of body cholesterol is produced by the liver, and 50% of this is reabsorbed back into the circulation via the small intestine.
Key Points
- Cholesterol is also synthesized in the adrenal glands and reproductive organs.
- Cholesterol is kept in balance by homeostatic mechanisms: higher dietary intake leads to reduced synthesis in the body.
- Dietary cholesterol is poorly absorbed; therefore, intake of cholesterol has little effect on blood cholesterol levels.
- Cholesterol forms part of the bile acids excreted from the liver.
- Crystallized cholesterol is the primary component of gallstones.
- Cholesterol in the colon is metabolized by colonic bacteria which convert it to a nonabsorbable sterol that is excreted in the feces.
Key Terms
- gallbladder: In vertebrates, a small organ that aids mainly in fat digestion and concentrates bile produced by the liver.
- cholesterol: A sterol lipid synthesized by the liver and transported in the bloodstream to the membranes of all animal cells; it plays a central role in many biochemical processes and, as a lipoprotein that coats the walls of blood vessels, is associated with cardiovascular disease.
- bile: A bitter brownish-yellow or greenish-yellow secretion produced by the liver, stored in the gall bladder, and discharged into the duodenum, where it aids the process of digestion.
EXAMPLES
Eggs are an excellent, low-calorie source of protein, but they also are fairly high in cholesterol, so, in the past, it was thought that eggs should be limited in the diet. However, as we now know that cholesterol is kept in balance in the body, and that food intake has little effect on blood cholesterol concentrations, it appears that eggs can be a healthy part of one’s diet.
About 20–25% of total daily cholesterol production occurs in the liver. Other sites of higher synthesis rates include the intestines, adrenal glands, and reproductive organs. Biosynthesis of cholesterol is directly regulated by the cholesterol levels present, although the homeostatic mechanisms involved are only partly understood. A higher intake from food leads to a net decrease in endogenous production, whereas lower intake from food has the opposite effect. However, most ingested cholesterol is esterified; esterified cholesterol is poorly absorbed. The body also compensates for any absorption of additional cholesterol by reducing cholesterol synthesis. For these reasons, cholesterol intake in food has little, if any, effect on total body cholesterol content or concentrations of cholesterol in the blood.

Cholesterol: A molecule of cholesterol consisting of four linked hydrocarbon rings forming the bulky steroid structure. There is a hydrocarbon tail linked to one end of the steroid and a hydroxyl group linked to the other end.
Cholesterol is recycled. The liver excretes it in a non-esterified form (via bile) into the digestive tract. Typically about 50% of the excreted cholesterol is reabsorbed by the small bowel back into the bloodstream.
Cholesterol is oxidized by the liver into a variety of bile acids. These, in turn, are conjugated with glycine, taurine, glucuronic acid, or sulfate. A mixture of conjugated and nonconjugated bile acids, along with cholesterol itself, is excreted from the liver into the bile. Approximately 95% of the bile acids are reabsorbed from the intestines, and the remainder is lost in the feces. The excretion and reabsorption of bile acids forms the basis of the enterohepatic circulation, which is essential for the digestion and absorption of dietary fats. Under certain circumstances, when more concentrated, as in the gallbladder, cholesterol crystallizes and is the major constituent of most gallstones. Lecithin and bilirubin gallstones also occur, but less frequently. Every day, up to one gram of cholesterol enters the colon. This cholesterol originates from the diet, bile, and desquamated intestinal cells; it can be metabolized by the colonic bacteria. Cholesterol is converted mainly into coprostanol, a nonabsorbable sterol that is excreted in the feces. A cholesterol-reducing bacterium origin has been isolated from human feces.
Selected factors implicated in cholesterol metabolism
| Factor | Full name | Function |
|---|---|---|
| ABCA1 | ATP binding cassette subfamily A member 1 | Cholesterol export protein; key factor in HDL biogenesis |
| ABCG1 | ATP binding cassette subfamily G member 1 | Cholesterol export protein |
| ABCG5 | ATP binding cassette subfamily G member 5 | Biliary sterol exporter |
| ABCG8 | ATP binding cassette subfamily G member 8 | Biliary sterol exporter |
| Apo | Apolipoprotein | The structural component of lipoproteins; stabilizes lipoproteins and is an interaction partner for cellular receptors |
| ATF4 | Activating transcription factor 4 | Downstream signaling protein of the UPR; activated by PERK |
| ATF6 | Activating transcription factor 6 | Downstream signaling protein of the UPR; mediates transcription of chaperones |
| GRP78 | Glucose-related protein 78 | ER chaperone |
| HDL | High-density lipoprotein | Cholesterol transport vehicle; main factor in RCT; transports cholesterol to the liver and steroidogenic tissues |
| HMG-CoAR | 3-hydroxy-3-methylglutaryl-CoA reductase | Rate-liming enzyme in cholesterol biosynthesis; target of statins |
| IDOL | Inducible degrader of the LDLR | Negative regulator of LDL uptake |
| INSIG | Insulin-induced gene | Blocks SREBP activation in response to high cellular cholesterol levels |
| IRE1 | Inositol-requiring enzyme 1 | Downstream signaling protein of the UPR; highly conserved among species |
| LDL | Low-density lipoprotein | Main cholesterol transport vehicle in humans |
| LDLR | Low-density lipoprotein receptor | Main receptor supplying cells with cholesterol |
| LXR | Liver X receptor | Positive regulator of cholesterol efflux and fatty acid synthesis |
| NRF1 | Nuclear respiratory factor 1; official gene name: NFE2L1 (nuclear factor, erythroid 2 like 1) | Transcription factor which is activated in response to high cellular cholesterol levels; positive regulator of cholesterol efflux |
| PCSK9 | Proprotein convertase subtilisin-like Kexin type 9 | Negative regulator of plasma LDL levels; mediates degradation of the LDLR |
| PERK | Protein kinase RNA-like ER kinase | Downstream signaling protein of the UPR; excess PERK-activation triggers apoptosis |
| RCT | Reverse cholesterol transport | Transport of excess peripheral cholesterol into the liver for disposal into the bile |
| S1P, S2P | Site- 1/2 protease | Golgi-resident proteases which activate transcription factors involved in cholesterol homeostasis (SREBP) and unfolded protein response (ATF6) |
| SCAP | SREBP cleavage-activating protein | Mediates transport of SREBP from the ER to the Golgi apparatus |
| SR-BI | Scavenger receptor class B member 1 | HDL receptor |
| SREBP | Sterol regulatory element-binding protein | Transcription factor which is activated in response to low cellular cholesterol levels; positive regulator of cholesterol uptake and synthesis |
| UPR | Unfolded protein response | Signaling cascade activated by ER stress; mediates adaptive mechanisms or apoptosis |
Regulation of Blood Cholesterol Levels
Cholesterol is transported through the blood by lipoproteins which direct cholesterol to where it is needed.
Key Points
There are several types of lipoproteins that are classified by their density or ratio of lipid to protein. The lower the protein, the less dense it is.
LDL lipoproteins contain the highest percentage of cholesterol and are the major carriers through the bloodstream.
LDL and its receptor form a vesicle which fuses with a lysosome that releases the cholesterol for use in membrane biosynthesis or cellular storage.
HDL lipoproteins transport cholesterol back to the liver for excretion or reprocessing.
High levels of HDL lipoproteins are associated with better health outcomes.
LDL lipoproteins contain the highest percentage of cholesterol and are the major carriers through the bloodstream.LDL and its receptor form a vesicle which fuses with a lysosome that releases the cholesterol to be used in membrane biosynthesis or to be stored in the cell.
When a cell has sufficient cholesterol LDL receptors are blocked, but when the cell is deficient, more LDL receptors are created.
If the system breaks down, LDL lipoproteins accumulate in the blood, are engulfed by macrophages, and may become trapped on the walls of blood vessels to form plaques.
HDL lipoproteins transport cholesterol back to the liver for excretion or reprocessing.
High levels of HDL lipoproteins are associated with better health outcomes.
Key Terms
- lysosome: An organelle found in all types of animal cells which contains a large range of digestive enzymes capable of splitting most biological macromolecules.
- lipoprotein: Any of a large group of complexes of protein and lipid with many biochemical functions.
- chylomicrons: Chylomicrons are one of the five major groups of lipoproteins (chylomicrons, VLDL, IDL, LDL, HDL) that enable fats and cholesterol to move within the water-based solution of the bloodstream.
EXAMPLES
A cholesterol test will measure your HDL, LDL, VLDL, and triglyceride levels. The ratio of HDL to LDL is a useful screening method to determine your possible risk for heart disease or stroke.
Blood Cholesterol Levels and Transport
Cholesterol is only slightly soluble in water; it can dissolve and travel in the water-based bloodstream at exceedingly small concentrations. Since cholesterol is insoluble in blood, it is transported in the circulatory system within lipoproteins, which are complex discoidal particles that have an exterior composed of amphiphilic proteins and lipids whose outward-facing surfaces are water-soluble and inward-facing surfaces are lipid-soluble; triglycerides and cholesterol esters are carried internally. Phospholipids and cholesterol, being amphipathic, are transported in the surface monolayer of the lipoprotein particle.

liposome structure: Lipid vesicles (or liposomes) are small, intracellular, membrane-enclosed bubbles of liquid within a cell. They are formed because of the properties of lipid membranes, where the hydrophobic chains are packed together so they are not in contact with the aqueous solution.
In addition to providing a soluble means for transporting cholesterol through the blood, lipoproteins have cell-targeting signals that direct the lipids they carry to certain tissues. For this reason, there are several types of lipoproteins within the blood called, in order of increasing density:
- chylomicrons
- very-low-density lipoprotein (VLDL)
- intermediate-density lipoprotein (IDL)
- low-density lipoprotein (LDL), and
- high-density lipoprotein (HDL).
The more lipid and less protein a lipoprotein has, the less dense it is. The cholesterol within all the various lipoproteins is identical, although some cholesterol is carried as the “free” alcohol and some is carried as fatty acyl esters referred to as cholesterol esters. However, the different lipoproteins contain apolipoproteins, which serve as ligands for specific receptors on cell membranes. In this way, the lipoprotein particles are molecular addresses that determine the start and endpoints for cholesterol transport.
Chylomicrons, the least dense type of cholesterol transport molecules, contain apolipoprotein B-48, apolipoprotein C, and apolipoprotein E in their shells. Chylomicrons are the transporters that carry fats from the intestine to muscle and other tissues that need fatty acids for energy or fat production. Cholesterol that is not used by muscles remains in more cholesterol-rich chylomicron remnants, which are taken up from here to the bloodstream by the liver.
VLDL molecules are produced by the liver and contain excess triacylglycerol and cholesterol that is not required by the liver for synthesis of bile acids. These molecules contain apolipoprotein B100 and apolipoprotein E in their shells. During transport in the bloodstream, the blood vessels cleave and absorb more triacylglycerol from IDL molecules, which contain an even higher percentage of cholesterol. The IDL molecules have two possible fates: half are metabolized by HTGL, taken up by the LDL receptor on the liver cell surfaces and the other half continue to lose triacylglycerols in the bloodstream until they form LDL molecules, which have the highest percentage of cholesterol within them.
LDL molecules, therefore, are the major carriers of cholesterol in the blood, and each one contains approximately 1,500 molecules of cholesterol ester. The shell of the LDL molecule contains just one molecule of apolipoprotein B100, which is recognized by the LDL receptor in peripheral tissues. Upon binding of apolipoprotein B100, many LDL receptors become localized in clathrin-coated pits. Both the LDL and its receptor are internalized by endocytosis to form a vesicle within the cell. The vesicle then fuses with a lysosome, which has an enzyme called lysosomal acid lipase that hydrolyzes the cholesterol esters. Now within the cell, the cholesterol can be used for membrane biosynthesis or esterified and stored within the cell, so as to not interfere with cell membranes.
Synthesis of the LDL receptor is regulated by SREBP, the same regulatory protein as was used to control synthesis of cholesterol de novo in response to cholesterol presence in the cell. When the cell has abundant cholesterol, LDL receptor synthesis is blocked so new cholesterol in the form of LDL molecules cannot be taken up. On the converse, more LDL receptors are made when the cell is deficient in cholesterol. When this system is deregulated, many LDL molecules appear in the blood without receptors on the peripheral tissues. These LDL molecules are oxidized and taken up by macrophages, which become engorged and form foam cells. These cells often become trapped in the walls of blood vessels and contribute to artherosclerotic plaque formation. Differences in cholesterol homeostasis affect the development of early atherosclerosis (carotid intima-media thickness). These plaques are the main causes of heart attacks, strokes, and other serious medical problems, leading to the association of so-called LDL cholesterol (actually a lipoprotein) with “bad” cholesterol.
Also, HDL particles are thought to transport cholesterol back to the liver for excretion or to other tissues that use cholesterol to synthesize hormones in a process known as reverse cholesterol transport (RCT). Having large numbers of large HDL particles correlates with better health outcomes. In contrast, having small numbers of large HDL particles is independently associated with atheromatous disease progression within the arteries.

Cholesterol Blood Tests: Reference ranges for blood tests, showing usual, as well as optimal, levels of HDL, LDL and total cholesterol in mass and molar concentrations, is found in orange color at right, that is, among the blood constituents with the highest concentration.
References
[bg_collapse view=”button-orange” color=”#4a4949″ expand_text=”Show More” collapse_text=”Show Less” ]
- https://www.ncbi.nlm.nih.gov/books/NBK22259/
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6132555/
- https://www.ncbi.nlm.nih.gov/books/NBK395580/
- https://www.ncbi.nlm.nih.gov/books/NBK470561/
- https://www.ncbi.nlm.nih.gov/books/NBK513326/
- https://www.ncbi.nlm.nih.gov/books/NBK544353/
- https://www.ncbi.nlm.nih.gov/books/NBK22259/
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6020734/
- https://www.ncbi.nlm.nih.gov/books/NBK22437/
- https://www.ncbi.nlm.nih.gov/books/NBK9903/
- https://www.ncbi.nlm.nih.gov/books/NBK525983/
- https://www.ncbi.nlm.nih.gov/books/NBK546690/
- https://www.ncbi.nlm.nih.gov/books/NBK326737/
- https://www.ncbi.nlm.nih.gov/books/NBK232461/
- https://www.ncbi.nlm.nih.gov/books/NBK209054/
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4224210/
- https://www.ncbi.nlm.nih.gov/books/NBK234933/
- https://www.ncbi.nlm.nih.gov/books/NBK279012/
- https://www.ncbi.nlm.nih.gov/books/NBK6824/
- https://www.ncbi.nlm.nih.gov/books/NBK21154/
- https://www.ncbi.nlm.nih.gov/books/NBK26820/
- https://www.ncbi.nlm.nih.gov/books/NBK224624/
- https://www.ncbi.nlm.nih.gov/books/NBK21475/
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3649104/
- https://www.ncbi.nlm.nih.gov/books/NBK248/
- https://www.ncbi.nlm.nih.gov/books/NBK234922/
- https://www.sciencedirect.com/topics/chemistry/metabolic-reaction
- https://www.sciencedirect.com/book/9780124177628/nutrient-metabolism#book-description
[/bg_collapse]
Visitor Rating: 5 Stars