Pulmonary embolism (PE) is a common illness which requires early diagnosis and treatment due to its association with high mortality and morbidity rates [rx–rx]. Significant advances have been achieved in the diagnosis and treatment of PE since venous thromboembolism (VTE) and its triad of contributing factors, including vascular endothelial damage, hypercoagulation and venous stasis, were first defined nearly 150 years ago by Rudolph Virchow, a German pathologist [rx].
Acute pulmonary embolism (APE) is the most serious clinical presentation of venous thromboembolism (VTE) with fatal pulmonary embolism (PE) being a common cause of sudden death (SD), usually resulting from a complication of deep venous thrombosis (DVT).
Pulmonary embolism (PE) is an acute and potentially fatal condition in which embolic material, usually a thrombus originating from one of the deep veins of the legs or pelvis, blocks one or more pulmonary arteries, causing impaired blood flow and increased pressure to the right cardiac ventricle. Pulmonary embolism and deep vein thrombosis are considered to be two manifestations of the same condition, venous thromboembolism, which is the third most common cardiovascular disorder in industrialized countries [rx,rx].
Pathophysiology
The results of acute PE symptoms are essentially hemodynamic and become significant when greater than 30–50% of the pulmonary artery bed is obstructed by emboli (pulmonary artery pressure > 30–40 mm Hg). Factors affecting the level of hemodynamic dysfunction include thrombus size, the diameter of the affected vessels, embolism type, platelet dysfunctions, neurohumoral substances released from the damaged endothelial and mast cells, and the patient’s previous cardiopulmonary reserve. Initially, pulmonary vascular resistance increases suddenly, due to the thrombus settling in the pulmonary vascular bed. The thrombus leads to an increase in pulmonary vascular resistance by narrowing the pulmonary vascular bed and vasospasm, and an imbalance in ventilation/perfusion (V/Q), by reducing pulmonary circulation. Increased pulmonary vascular resistance and elevated pulmonary artery pressure may lead to a reduction in right ventricle dilatation and subsequent decrease in blood outflow volume and systemic hypotension [rx].
The expected pathological consequences in the lung tissue, depending on the obstructed vessel, include alveolar dead space respiration, hyperventilation caused by hypoxemia, alveolar hypocapnia, bronchoconstriction, alveolar collapse, and atelectasis due to reduction in surfactant production. Therefore, dynamic compliance is reduced and the alveolar-arterial gradient is increased in the lungs [rx].
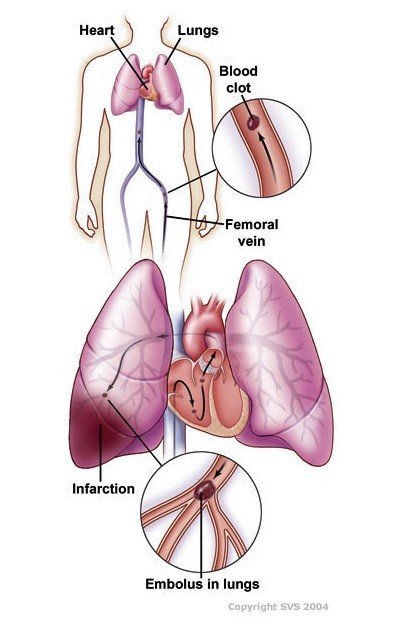
Causes of Pulmonary Embolism
- Injury or damage – Injuries like bone fractures or muscle tears can cause damage to blood vessels, leading to clots.
- Inactivity – During long periods of inactivity, gravity causes blood to stagnate in the lowest areas of your body, which may lead to a blood clot. This could occur if you’re sitting for a lengthy trip or if you’re lying in bed recovering from an illness.
- Medical conditions – Some health conditions cause blood to clot too easily, which can lead to pulmonary embolism. Treatments for medical conditions, such as surgery or chemotherapy for cancer, can also cause blood clots.
[stextbox id=’custom’]
Potential causes of increased cardiac troponin levels
| Cardiac disease with a potential increase in troponin levels | Noncardiac disease with a potential increase in troponin levels |
|---|---|
| Pulmonary embolism, pulmonary hypertension | Infiltrative diseases such as sarcoidosis, amyloidosis, scleroderma, hemochromatosis |
| Hypertrophic cardiomyopathy | Acute neurological conditions including stroke and subarachnoidal hemorrhage |
| Aortic dissection Aortic valve disease Inflammatory diseases – myocarditis, pericarditis, endocarditis Cardiac contusion Acute and chronic advanced-stage heart failure Hypertensive crisis Tachyarrhythmia, bradyarrhythmia Cardioversion, cardiac pacing, endomyocardial biopsy, post ablation states Takotsubo syndrome |
Acute and chronic renal failure Burns involving more than 30% of body surface area Rhabdomyolysis Toxic effects of drugs – adriamycin, 5-fluorouracil, Herceptin, snake toxins Hypothyroidism Critically ill patients with sepsis, respiratory failure |
Adapted from reference [rx]
Risk factors of venous thromboembolism, according to the British Thoracic Society, 2003
[/stextbox]
Major risk factors
- Postoperative states: Major abdominal/pelvic surgery, hip/knee joint replacement, postoperative intensive care
- Obstetrics: Late pregnancy, Caesarian section, puerperium
- Lower limb affections: Fractures, extensive varicosities
- Malignancies: Abdominal/pelvic, advanced/metastatic stage
- Limited mobility: Hospitalization, geriatric care
- Miscellaneous: History of previous venous thromboembolism
Minor
- Cardiovascular – Congenital heart disease, heart failure, hypertension, superficial venous thrombosis, central venous catheter
- Humoral – Estrogen use: oral contraception, hormone replacement therapy
- Miscellaneous – Chronic obstructive lung disease, neurological impairment, latent malignancy, thrombotic defects, long-distance travel in the sitting position, obesity
- Other – Inflammatory bowel disease, nephrotic syndrome, chronic dialysis, myeloproliferative disease, paroxysmal nocturnal hemoglobinuria
Symptoms of Pulmonary Embolism
- Shortness of breath – This symptom typically appears suddenly and always gets worse with exertion.
- Chest pain – You may feel like you’re having a heart attack. The pain may become worse when you breathe deeply (pleurisy), cough, eat, bend or stoop. The pain will get worse with exertion but won’t go away when you rest.
- Cough – The cough may produce bloody or blood-streaked sputum.
- Swelling of the affected leg or arm.
- Leg pain or tenderness that may only occur when you are standing or walking.
- Increased warmth in the swollen or painful area of the affected leg or arm.
- Redness or discoloration of your skin.
- Enlargement of superficial veins in the affected leg or arm.
- SD (sudden cardiac arrest; SCA)
- Similar to acute respiratory distress syndrome (ARDS)
- Typical respiratory failure (hypoxia and hypocapnia)
- Asthmatic crisis-like syndrome
- Fever syndrome with or without pleuropneumonia (with or without pleural effusion)
- Acute right heart failure/shock/hypotension (often with epigastric pain)
- Left heart failure (with pulmonary congestion)
- Chest pain similar to pleuritic syndrome with or without hemoptysis (with or without effusion)
- Similar to acute coronary syndrome (ACS) (with or without chest pain)
- PE with paradoxical embolism (with corresponding clinical picture due to embolization site and resulting in AMI, stroke, flank pain [due to acute splenic infarction or acute renal infarction], acute abdomen, and upper or lower extremity embolism. In all these paradoxical embolism conditions, the main symptom may be systemic or not)
- Syncope
- Complete atrioventricular (AV) block with idioventricular rhythm
- Persistent or paroxysmal atrial fibrillation (AF), atrial flutter, atrial tachycardia, paroxysmal supraventricular tachycardia (PSVT)
- DVT and silent PE
- Platypnea-orthodeoxia
- Abdominal pain without abdomen acute
- Delirium
- Swelling of a leg or arm
- Leg pain or tenderness when you’re standing or walking
- A swollen leg or arm that feels warmer than normal
- Red or discolored skin in the affected arm or leg
- Veins in your arm or leg that are larger than normal
- Leg pain or swelling, or both, usually in the calf
- Clammy or discolored skin (cyanosis)
- Fever
- Excessive sweating
- Rapid or irregular heartbeat
- Lightheadedness or dizziness
- Pain in your back
- Much more sweating than usual
- A lightheaded feeling, or passing out
- Blue lips or nails
Diagnosis of Pulmonary Embolism
There are additional prediction rules for PE, such as the Geneva rule. More importantly, the use of any rule is associated with a reduction in recurrent thromboembolism.[rx]
The Wells score[rx]
- Clinically suspected DVT — 3.0 points
- An alternative diagnosis is less likely than PE — 3.0 points
- Tachycardia (heart rate > 100) — 1.5 points
- Immobilization (≥ 3d)/surgery in previous four weeks — 1.5 points
- History of DVT or PE — 1.5 points
- Hemoptysis — 1.0 points
- Malignancy (with treatment within six months) or palliative — 1.0 points
Traditional interpretation[rx][rx][rx]
- Score >6.0 — High (probability 59% based on pooled data)[rx]
- Score 2.0 to 6.0 — Moderate (probability 29% based on pooled data)[rx]
- Score <2.0 — Low (probability 15% based on pooled data)[rx]
Alternative interpretation[rx][rx]
- Score > 4 — PE likely. Consider diagnostic imaging.
- Score 4 or less — PE unlikely. Consider D-dimer to rule out PE.
Recommendations for a diagnostic algorithm were published by the PIOPED investigators; however, these recommendations do not reflect research using 64 slice MDCT.[rx] These investigators recommended:
- Low clinical probability – If negative D-dimer, PE is excluded. If positive D-dimer, obtain MDCT and based treatment on results.
- Moderate clinical probability – If negative D-dimer, PE is excluded. However, the authors were not concerned that a negative MDCT with negative D-dimer in this setting has a 5% probability of being false. Presumably, the 5% error rate will fall as 64 slice MDCT is more commonly used. If positive D-dimer, obtain MDCT and based treatment on results.
- High clinical probability – Proceed to MDCT. If positive, treat, if negative, more tests are needed to exclude PE. A D-dimer of less than 750 ug/L does not rule out PE in those who are at high risk.[rx]
- Pulmonary embolism rule-out criteria – The pulmonary embolism rule-out criteria (PERC) helps assess people in whom pulmonary embolism is suspected, but unlikely. Unlike the Wells score and Geneva score, which are clinical prediction rules intended to risk stratify people with suspected PE, the PERC rule is designed to rule out the risk of PE in people when the physician has already stratified them into a low-risk category.
- Chest X-ray – Pulmonary embolism should also be suspected in patients who are found to have hypoxemia through arterial blood gas analysis but have normal chest X-rays and no evidence of airway obstruction. Although there are nonspecific chest X-ray findings associated with PE, chest x-rays are useful in excluding other causes of dyspnea and chest pain.
- Electrocardiography and echocardiography – Although ECG changes are not specific to PE, they are associated with right ventricle loading. Electrocardiography findings indicating right ventricle loading may include sinus tachycardia, atrial fibrillation, atrial extrasystoles, T wave inversion in V1-V4 derivations or a QR pattern in a V1 derivation, acute right ventricle dysfunction (classical S1Q3T3 pattern), an S wave deeper than 1.5 mm in DI or aVL derivations, a Q wave in DIII and aVF, voltage reduction in extremity derivations, and an incomplete or complete right bundle branch block.
- Computerized tomography pulmonary angiogram – In recent years, computerized tomography pulmonary angiogram (CTPA) has become the preferred diagnostic imaging method for the detection of PE in large and segmental arteries. Computerized tomography pulmonary angiogram has replaced ventilation/perfusion (V/Q) scintigraphy and pulmonary angiography, due to its ability to assess the pulmonary parenchyma and pleural space, as well as to precisely demonstrate the thrombus within the lumen [rx, rx]. In the Prospective Investigation of Pulmonary Embolism II (PIOPED II), a multi-center study involving 774 patients, multi-detector CTPA was reported to have a sensitivity of 83% and specificity of 96% [rx].
- Pulmonary angiography – Today, pulmonary angiography still remains the gold standard for diagnosis of PE by visualizing a filling defect or blunting in a pulmonary arterial branch. Subsegmental arteries of up to 1–2 mm in diameter can be assessed by pulmonary angiography, which is invasive, expensive, not available in every center, and associated with several risks.
- Magnetic resonance images – Magnetic resonance imaging (MRI) can be used as an alternative to CTPA for patients with renal failure, pregnant women and contraindication to administration of iodinated contrast agents. Magnetic resonance images can be used for a confident diagnosis of pulmonary embolism from the main pulmonary artery through the segmental branches of subsegmental pulmonary arteries [rx].
- Lower extremity ultrasonography – Pulmonary embolism mostly originates in the deep veins of the lower extremities [rx]. Positive findings observed during lower extremity ultrasonography in patients suspected of PE facilitate the decision to initiate anticoagulant treatment without further diagnostic evaluation. Although colored Doppler ultrasonography is not the gold standard for diagnosis of DVT, it is commonly used as such. Acquisition of lower extremity sections and visualization of the iliac veins and inferior vena cava, following the acquisition of thoracic sections during CTPA evaluation, increases sensitivity by providing additional information regarding DVT [rx].
- Ventilation/perfusion scintigraphy (V/Q scintigraphy) – CTPA undoubtedly plays an increasing role in the diagnosis of PE [rx]. Despite being associated with high rates of inconclusive results, V/Q scintigraphy can also be used as a reliable imaging method in centers lacking CTPA, and for patients with a high clinical probability of PE but with a contrast medium allergy or renal failure. Anticoagulant treatment can be safely postponed in patients with normal perfusion scintigraphy results [rx].
- Serum D-dimer level – D-dimer, a fibrin degradation product, should be used to exclude PE in patients with low or intermediate probability results. Increased levels of serum D-dimer may be helpful in the diagnosis of PE, but serum D-dimer specificity is rather low since levels can also be elevated in a variety of conditions. The low specificity can be explained given that serum D-dimer level is specific for fibrin, not for PE. The most sensitive methods for measuring serum D-dimer levels are ELISA and the turbidimetric test. Serum D-dimer’s specificity for suspected PE is reduced by age and may be decreased to ≤ 10% after the age of 80 years [rx]. The multi-center Prolong study suggested that serum D-dimer level might be useful in treatment monitorization of patients with definite PE [rx].
- Cardiac troponins – Cardiac troponins may be elevated when right coronary artery circulation is reduced due to acute right-sided heart failure and in right ventricle infarction. Several studies have reported that elevated cardiac troponins are associated with poor prognosis for patients with PE [rx, rx].
- Brain natriuretic peptide – Brain natriuretic peptide (BNP) and N-terminal proBNP (NT-proBNP), a right ventricular dysfunction marker, released due to increased myocardial strain, were found to have prognostic significance as indicators of right ventricle dysfunction [rx].
- Duplex venous ultrasound – This test uses radio waves to visualize the flow of blood and to check for blood clots in your legs.
- Venography – This is a specialized X-ray of the veins of your legs.
Cardiac biomarkers
- Troponin – Troponin-T or -I can be raised in acute PE secondary to myocyte injury in the right ventricle, which correlates with the presence of RV dysfunction, cardiogenic shock and in-hospital mortality [rx, rx]. Raised troponin usually indicates massive or submassive PE, although it can be elevated in a patient with small PE [rx, rx–rx].
- Unliked-dimer troponin – cannot be used to rule out PE in the acute clinical setting, but can serve as a clue to the diagnosis of PE, and can be used in risk stratification of PE to identify low-risk patients with PE who can be treated as outpatients, especially if combined with other tests.
- BNP – BNP levels rise in response to stretch and/or increased ventricular pressure. Elevated levels of BNP are associated with RV dysfunction and RV failure, and it can be used to assess RV function in patients with PE. Indeed, a high BNP level has been shown to be associated with adverse outcome of PE [rx, rx, rx].
- Echocardiography – Echocardiographic abnormalities observed in PE include RV dysfunction and tricuspid regurgitation [rx–rx]. Regional wall motion abnormalities that spare the RV apex (McConnell’s sign) were found to be very suggestive of PE, with 77% sensitivity and 95% specificity [rx].
- Transesophageal echocardiography – has been used successfully to detect PE in the right heart and main pulmonary artery [rx], echocardiography has low specificity for PE and is not normally used as an initial screening test for this condition. However, echocardiography can be very useful in evaluating the severity of PE, especially if thrombolytic therapy is contemplated [rx, rx, rx, rx].
Treatment of Pulmonary Embolism
-
Low-molecular-weight heparin or fondaparinux for 5 days or until INR is greater than 2 for 24 hours (unfractionated heparin for patients with renal failure and increased risk of bleeding)
-
Vitamin K analogs for 3 months
-
In patients with cancer, consider anticoagulation for 6 months with low-molecular-weight heparin
-
In patients with unprovoked DVT consider vitamin K analogs beyond 3 months
-
Rivaroxaban is an oral factor Xa inhibitor which has recently been approved by the FDA and NICE and is attractive because there is no need for regular INR monitoring
Initial treatment
- Studies have shown that unfractionated heparin (UFH) – low molecular weight heparin (LMWH), and fondaparinux can be used for the treatment of PE. Although PE and DVT appear to be two different manifestations of the same disease, PE is still mostly treated by hospitalization. Since high-risk PE is associated with an early death rate (inpatient or initial 30-day mortality) of > 15%, hospitalization is required.
- According to the current guidelines – the mortality rate in the low-risk PE group is < 1%, and hospitalization may not be necessary for all patients in this group [rx]. However, due to a lack of consensus, hospitalization is still recommended for treatment initiation.
- Hemodynamic dysfunction – should be corrected in high-risk PE patients presenting with shock and hypotension. Oxygen and fluid support should be provided, and UFH should be initiated without delay. Following an initial intravenous dose (40–80 IU/kg or 5000 IU), a continuous infusion should be administered (in 5% dextrose solution at a rate of 14–18 IU/kg/h or 1300 IU/h).
- Unfractionated heparin monitorization – should be performed to keep the target aPTT of the patient 1.5–2.5 times higher than the baseline aPTT.
- Hemorrhage – transient benign thrombocytopenia in the first 2–5 days, heparin-related immune thrombocytopenia between 5 and 15 days, hypersensitivity reactions including urticaria, angioedema, anaphylaxis hyperkalemia due to reduction of aldosterone, or osteoporosis after use for 1 month or longer may be observed due to UFH treatment.
- Unfractionated heparin – can be used safely during pregnancy and lactation. In cases of heparin resistance, where the aPTT remains below the therapeutic level despite a treatment dosage of > 35 000 IU/day, anti-factor Xa activity should be investigated, and LMWH treatment should be administered.
- If thrombolytic treatment is considered – UFH can be stopped and streptokinase (loading dose of 250 000 IU in 30 min, and maintenance dose of 100 000 IU/h within 12–24 h, rapid treatment 1.5 million IU/2 h), urokinase (loading dose of 4400 IU/kg in 10 min, and maintenance dose of 4400 IU/kg/h within 12–24 h, rapid treatment 3 million IU/2 h) or recombinant tissue plasminogen activator (100 mg every 2 h) can be administered [53]. Thrombolytic treatment should not be administered to patients who had a tumor or experienced bleeding, hemorrhagic stroke, or ischemic central nervous system injury within the last 6 months. Patients who experienced trauma or underwent a surgical procedure within the last 3 months should be similarly excluded.
- apixaban, dabigatran, edoxaban or rivaroxaban– [rx]- Of these, apixaban and rivaroxaban do not require ‘bridging’ therapy with heparin, so are better suited for initiation of therapy in ED patients. Compared with long-term warfarin therapy, these DOC agents are associated with similar rates of recurrent PE but slightly lower rates of treatment-associated hemorrhage, in particular, intracranial hemorrhage.
- Dabigatran – is a direct thrombin (factor II) inhibitor approved for the treatment of acute VTE after initial treatment with heparin [rx]. Apixaban, edoxaban, and rivaroxaban are factor Xa inhibitors and approved for the treatment of VTE. However, while apixaban and rivaroxaban are approved as monotherapy, edoxaban requires a heparin bridge similar to dabigatran. The initiation dose of apixaban is 10 mg twice daily for 7 days followed by 5 mg twice daily after that period. The initiation dose of rivaroxaban is 15 mg orally, twice daily, followed by 20 mg daily after 21 days. All of the DOACs should be used with caution in patients with severe liver or kidney disease, and studies of high-risk subgroups of patients (e.g., antiphospholipid antibody) are limited.
- Alteplase (tPA) and tenecteplase – are two thrombolytic agents that can be used in the treatment of PE. Only alteplase is US FDA approved for the treatment of massive PE. Due to its short half-life of 4 to 6 minutes, alteplase requires a continuous IV infusion. An initial bolus of 15 mg followed by an additional 85 mg over 2 hours is standard dosing. For hemodynamically unstable patients, this relatively lengthy 2 hours infusion may be too slow and impractical. Here tenecteplase offers an alternative, with a longer 20 to 24 minutes half-life that is delivered in a single, weight-based bolus dose. Tenecteplase, however, is not currently FDA approved for VTE treatment, although it was used in two major randomized trials of PE thrombolysis [rx,rx].
- Clot removal – A thin tube called a catheter will suction large clots out of your artery. It isn’t an entirely effective method because of the difficulty involved, so it’s not always a preferred method of treatment.
- Open surgery – Doctors use open surgery only in emergency situations when a person is in shock or medications aren’t working to break up the clot.
- Vein filter. A catheter can also be used to position a filter in the body’s main vein — called the inferior vena cava — that leads from your legs to the right side of your heart. This filter can help keep clots from being carried into your lungs. This procedure is typically reserved for people who can’t take anticoagulant drugs or when anticoagulant drugs don’t work well enough or fast enough. Some filters can be removed when they are no longer needed.
- Clot busters – Clot-busters is one way to immediately remove a clot. Clot busters are taken intravenously, through an IV in the arm (systemic thrombolysis) or through a long catheter (thin tube) that delivers them directly to a clot in the lung (catheter-directed thrombolysis).
Prevention
Preventing clots in the deep veins in your legs (deep vein thrombosis) will help prevent pulmonary embolism. For this reason, most hospitals are aggressive about taking measures to prevent blood clots, including:
- Blood thinners (anticoagulants) – These medications are often given to people at risk of clots before and after an operation — as well as to people admitted to the hospital with a heart attack, stroke or complications of cancer.
- Compression stockings – Compression stockings steadily squeeze your legs, helping your veins and leg muscles move blood more efficiently. They offer a safe, simple and inexpensive way to keep blood from stagnating during and after general surgery.
- Leg elevation – Elevating your legs when possible and during the night also can be very effective. Raise the bottom of your bed 4 to 6 inches with blocks or books.
- Physical activity – Moving as soon as possible after surgery can help prevent pulmonary embolism and hasten recovery overall. This is one of the main reasons your nurse may push you to get up, even on your day of surgery, and walk despite the pain at the site of your surgical incision.
- Pneumatic compression – This treatment uses thigh-high or calf-high cuffs that automatically inflate with air and deflate every few minutes to massage and squeeze the veins in your legs and improve blood flow.
- Drink plenty of fluids – Water is the best liquid for preventing dehydration, which can contribute to the development of blood clots. Avoid alcohol, which contributes to fluid loss.
- Take a break from sitting – Move around the airplane cabin once an hour or so. If you’re driving, stop every hour and walk around the car a couple of times. Do a few deep knee bends.
- Fidget in your seat – Flex your ankles every 15 to 30 minutes.
- Wear support stockings – Your doctor may recommend these to help promote circulation and fluid movement in your legs. Compression stockings are available in a range of stylish colors and textures. There are even devices, called stocking butlers, to help you put on the stockings.
- Drink plenty of fluids. If you can’t drink, you’ll be given fluids through a drip.
- Get up and start moving about as soon as you can after an operation or illness.
- Wear compression stockings to help your circulation.
- Use an intermittent pneumatic compression device. This is an inflatable cuff wrapped around your leg or foot and an electrical pump that inflates it, squeezing your deep veins.
- Have anticoagulant medicines such as heparin.
References
[bg_collapse view=”button-orange” color=”#4a4949″ expand_text=”Show More” collapse_text=”Show Less” ]
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3665123/
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5940642/
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3878229/
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5065342/
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5065342/
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3718593/
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3718593/
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4107241/
- https://en.wikipedia.org/wiki/Pulmonary_embolism
- https://breathe.ersjournals.com/content/7/4/315
- http://www.bloodjournal.org/content/121/22/4443
- https://www.amjmed.com/article/S0002-9343(06)00783-2/fulltext
- http://www.bloodjournal.org/content/125/12/1877
- https://www.bmj.com/content/340/bmj.c1421.full
- https://jintensivecare.biomedcentral.com/articles/10.1186/s40560-018-0286-8
- https://www.wjgnet.com/2218-6255/full/v9/i3/30.htm
- https://emcrit.org/emcrit/fibrinolysis-in-pulmonary-embolism/
- https://www.mayoclinic.org/diseases-conditions/pulmonary-embolism/diagnosis-treatment/drc-20354653
- https://www.nhs.uk/conditions/pulmonary-embolism/
- https://www.webmd.com/lung/pulmonary-embolism-symptoms
- https://www.healthline.com/health/pulmonary-embolus
- https://medlineplus.gov/pulmonaryembolism.html
- https://vascular.org/patient-resources/vascular-conditions/pulmonary-embolism
- https://www.bupa.co.uk/health-information/heart-blood-circulation/pulmonary-embolism
- https://www.cedars-sinai.org/health-library/diseases-and-conditions/p/pulmonary-embolism.html
[/bg_collapse]

Visitor Rating: 5 Stars
Visitor Rating: 5 Stars